The biodegradability you need and the performance you want
Exxal™ alcohols are isomeric branched, primary alcohols that contain both even- and odd-numbered hydrocarbon chains, ranging from C8 to C13. In general, there are two main types of structures in industrial alcohols: Linear (or straight-chain) molecules; and Branched chain alcohols.
Our high-purity Exxal™ alcohols exhibit reactivity typical of higher primary alcohols. Thanks to their branched structure, Exxal alcohols offer many performance advantages to ethoxylates compared to linear alcohol-based ones. These advantages include:
- Low pour point; Biodegradability*
- Superior wetting power of derived surfactants
- Powerful solvency in coatings and inks, fully soluble in hydrocarbon fluids while enhancing their solvency power
- Wide viscosity range in synlubes
*All of our alcohols and their ethoxylates meet the OECD 301F readily biodegradable threshold for isomeric mixtures
| High performance > |
Biodegradability > |
Global supply > |
Performance properties you want
| Property | Units | Method | Principle |
|---|---|---|---|
| Cloud point in water | °C | CI-TM1 | Visual |
| Surface tension lowering (maximum) |
mN/m | CI-TM8 | Du Noüy ring |
| Occurence of gel phase at defined concentration and temperature |
wt% surfactant |
CI-TM5 | Rheometry |
| Wetting time in water | seconds | CI-TM3 | Draves test (cotton skein, based on AATCC method) |
| Foam height | mm | CI-TM6 | Dynamic foam analyser |
| Time to reach 40mN/m (20°C, 0.1 g/l in ,water) |
ms | CI-TM7 | Dynamic surface tension, maximum bubble pressure |
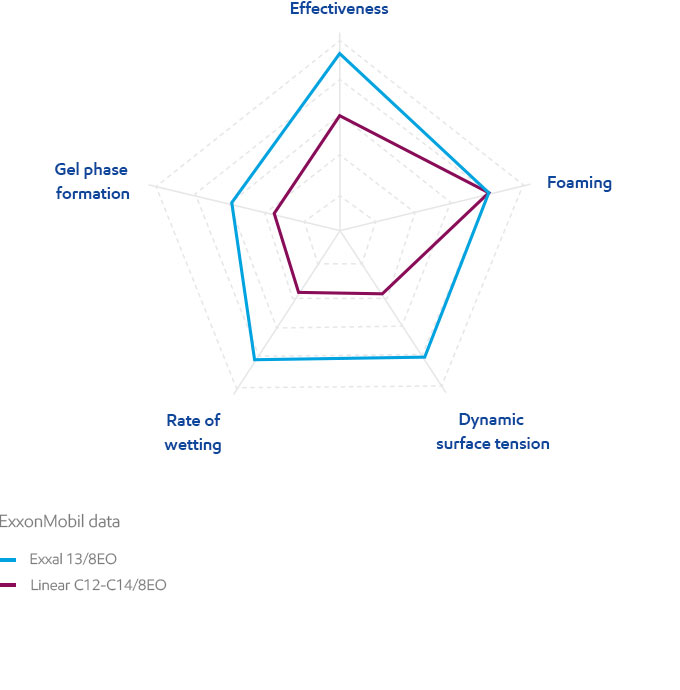
Effectiveness
A surfactant lowers the surface tension of water or the interfacial tension between water and organic liquids by adsorbing at the interface. Surfactant effectiveness is quantified by the lowest surface tension achievable, i.e. the surface tension at and beyond the CMC (Critical Micelle Concentration). Surface tension is measured by classical methods such as the Du Noüy ring. Exxal branched alcohol ethoxylates typically provide lower minimum surface tension values, but higher CMCs than the linear equivalents.
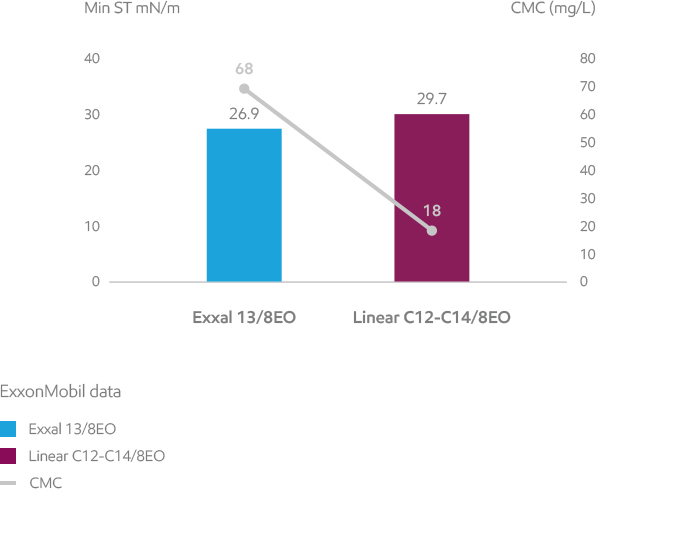
Exxal branched alcohol ethoxylates are effective surfactants and can help to reach lower minimum surface tension in water solutions.
Dynamic surface tension
The rate at which equilibrium surface tension is reached is also very important for a number of high-speed industrial processes. Typically, this can be expressed by the time required to reach a defined equilibrium surface tension (here 40 mN/m) for a fixed surfactant concentration (here 0.1 g/L) in water at 20°C. Branched alcohol ethoxylates need the least amount of time to reach the desired surface tension. Dynamic surface tension is measured by maximum bubble pressure.

Exxal branched alcohol ethoxylates need less time to reach the desired surface tension than linear based ethoxylates.
Foaming is a typical effect of surfactant solutions. It may or may not be desired, depending on the end-use. While the initial foam height is relatively similar for all branched ethoxylates, branched alcohols tend to have lower foam stability, which is generally an advantage in industrial surfactants. In the test, foam is generated by a gas flow under controlled conditions.
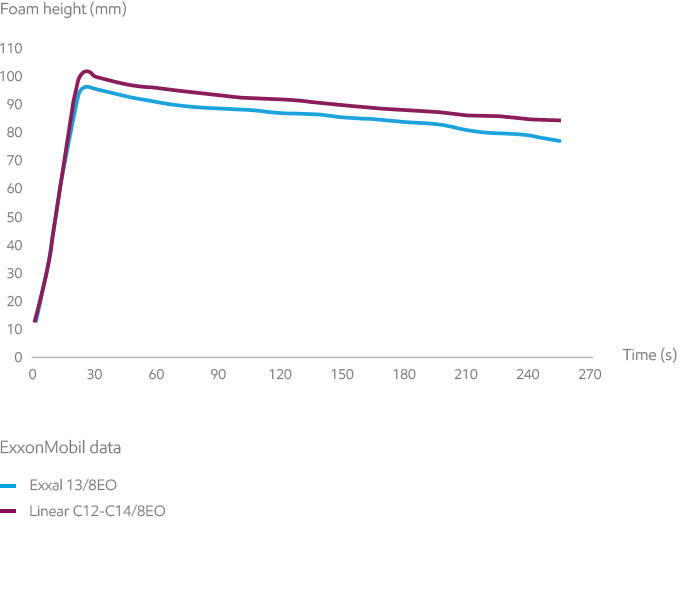
Exxal branched alcohol ethoxylates tend to have lower foam stability, which is generally an advantage in industrial surfactants.
Wetting of surfaces is essential for many surfactant applications. The rate of wetting can impact the efficiency of your process, both in speed and evenness of application. It refers to the time required to wet a standard cotton skein by a 1g/L surfactant solution (Draves test). There is a significant performance advantage for Exxal branched alcohol ethoxylates in industrial surfactants. Wetting time is significantly reduced from 12 to 4 seconds: 3 times lower than comparable linear alcohol ethoxylates. This results in lower processing times in applications like fast textile processing. Similarly, wetting performance leads to advantages in crop applications when active ingredients need to be quickly applied on surfaces.
The rate of wetting is measured by the Draves test.
The wetting time of ethoxylates based on Exxal branched alcohols is significantly lower than comparable linear alcohol based ethoxylates.
Gel phases make product handling more difficult and are generally avoided in industrial applications. Exxal branched alcohol ethoxylates tend to form fewer gel phases in water solutions than linear alcohols of comparable molecular weight. Due to this, solutions using Exxal based ethoxylates remain fluid, providing an important performance advantage for formulators or end users by improving product handling ability.

Ethoxylates based on Exxal branched alcohols tend to form fewer gel phases than linear ethoxylates.
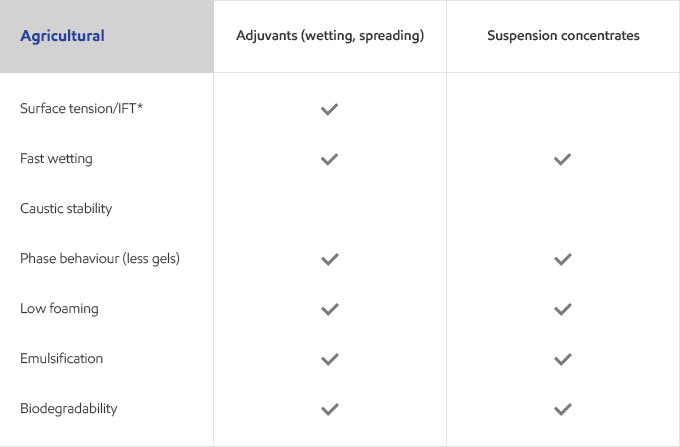
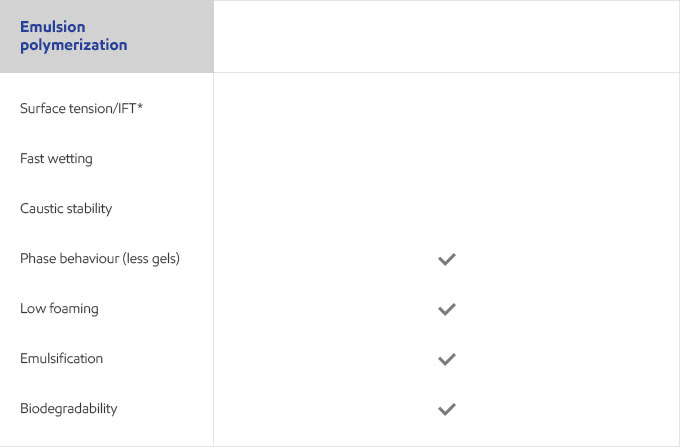
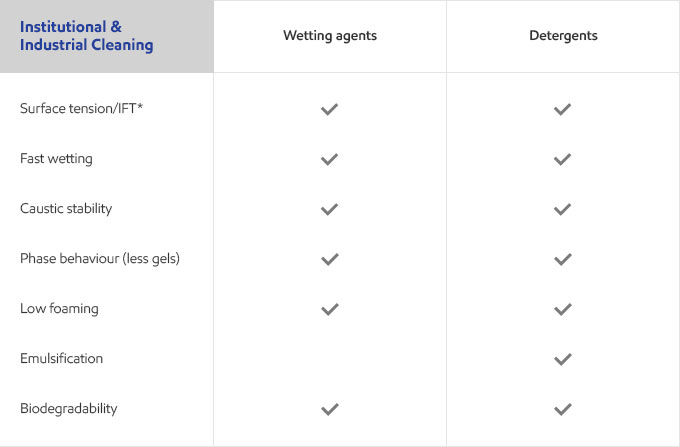
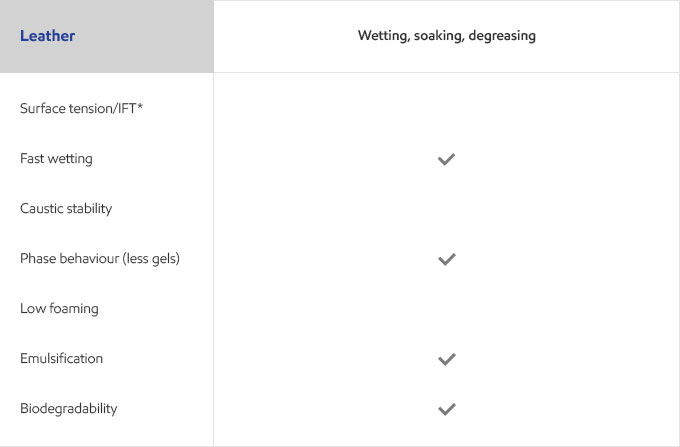
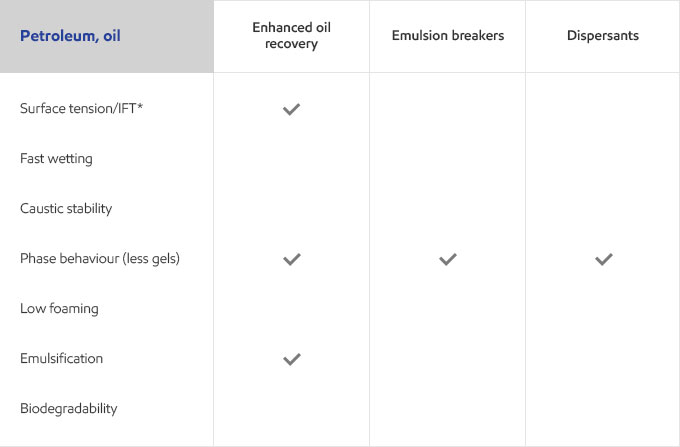
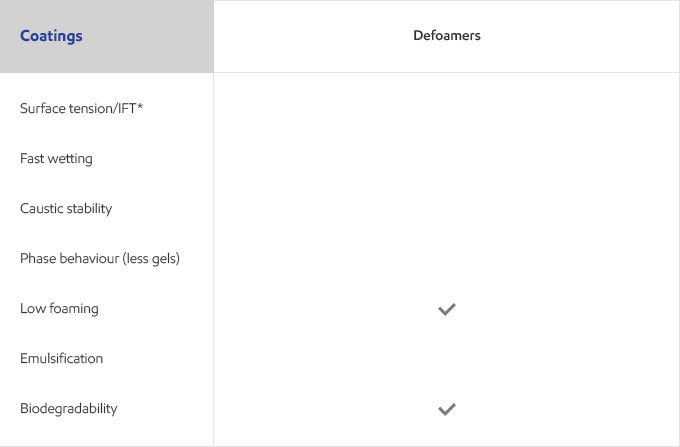
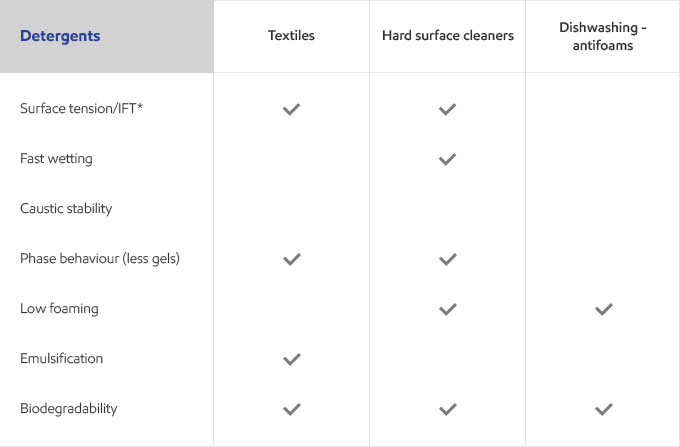
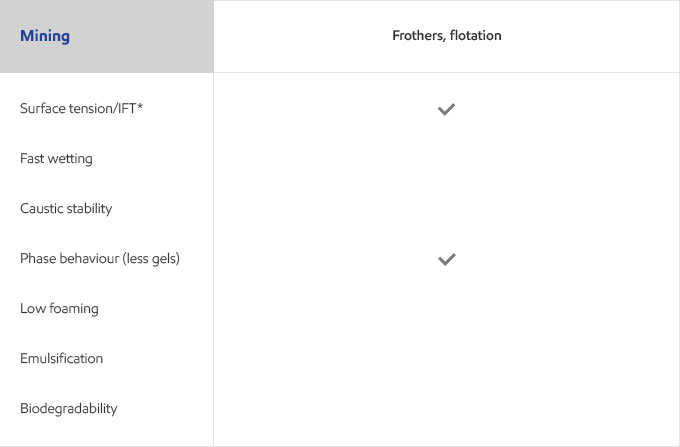
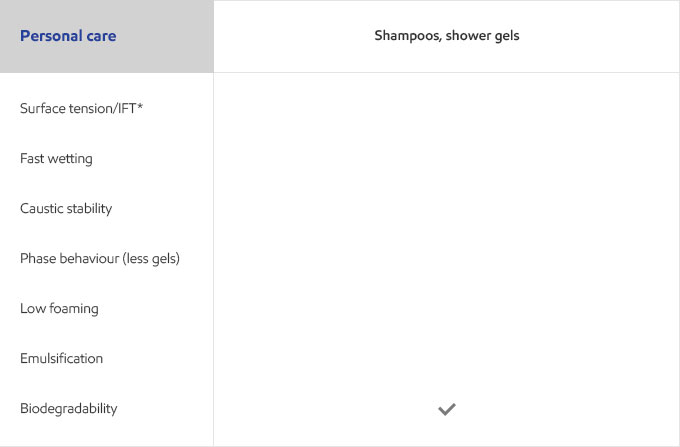
Whether your business is focused on textile, agricultural, personal care or detergents, you need surfactants that meet the specific needs of your customers and their end users. Exxal branched alcohols can provide a benefit to address those needs in many different surfactant applications.
Select your industry to get an overview of the benefits Exxal branched alcohols can have for your formulations.
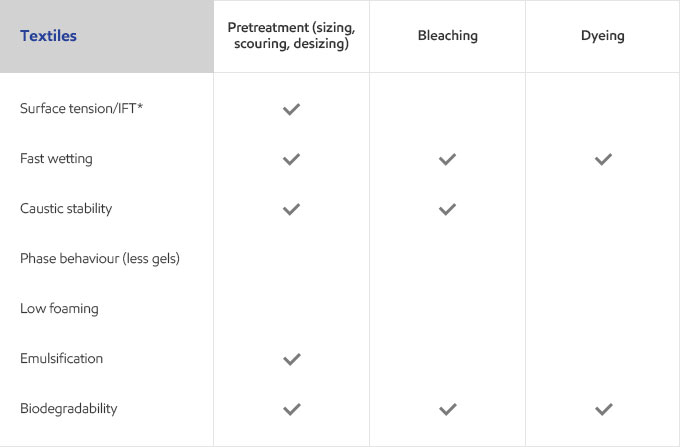
Get the best of both worlds
Are branched alcohol ethoxylates biodegradable? In short: yes!
Despite all the advantages of ethoxylates based on Exxal branched alcohols, such as superior rate of wetting and less gel phases, some manufacturers are reluctant to use branched alcohols due to the perception that branched alcohol ethoxylates do not biodegrade. ExxonMobil has used objective criteria and recognized test methods to show that Exxal branched alcohols and their ethoxylates do readily biodegrade.
One of the EPA and EU approved test methods is the OECD 301F manometric respirometry test that assesses “ultimate” biodegradation, which is the breakdown of the substance by microorganisms, resulting in the production of carbon dioxide, water, mineral salts, and new biomass. The criterion to “pass” as readily biodegradable in the 301F test is to reach 60% degradation in 28 days (for monoconstituent substances this would need to be achieved within a “10-day window”).
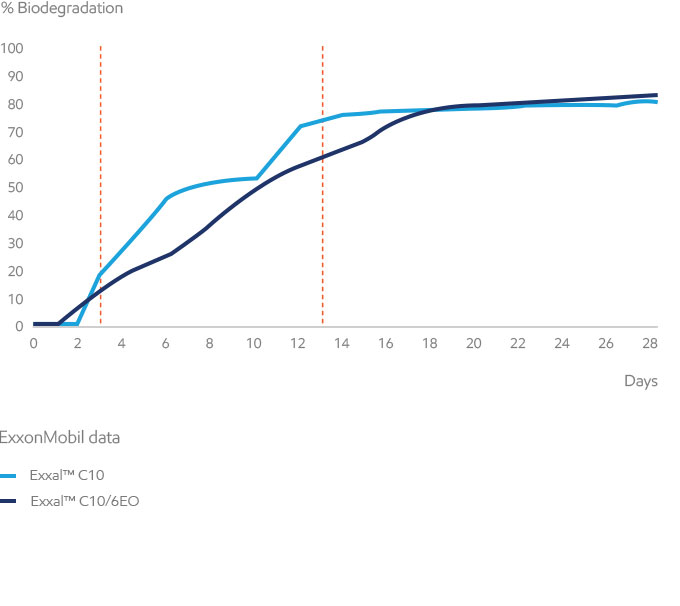
All of our alcohols and their ethoxylates meet the OECD readily biodegradable threshold for isomeric mixtures, and most of our alcohol ethoxylates also meet the “10-day window”. According to OECD guidance, the 10-day window should not be applied to isomeric mixtures of similar types of chemicals (e.g. surfactants made from Exxal branched alcohols). However, this requirement may be desired for certification with established eco-labelling programs.
Today, ExxonMobil offers six biodegradable Exxal branched alcohols that can be used to make a wide range of regulatory compliant biodegradable non-ionic surfactants (ethoxylates). Exxal branched alcohols can help fulfill the demand for a biodegradable surfactant that meets regulatory and voluntary standards – without compromising on the quality of your formulation.
ExxonMobil has a diverse alcohol grade slate that meets the standards

As your global partner, we are where you are
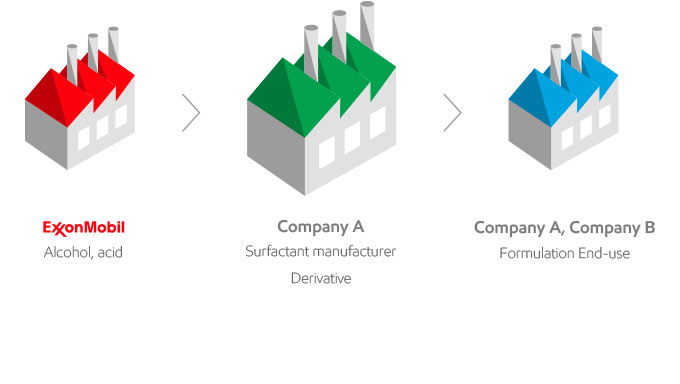
ExxonMobil is a supplier of raw materials to the surfactant industry. Because we are not integrated in the production of surfactants; we are working for and with you. This is what you can expect from working with ExxonMobil: a commitment to quality, innovation and the highest standards.
Tap into our expert environmental resources
Staying up to date with the latest environmental requirements is a key requirement in the surfactant industry. The scientific experts at ExxonMobil Biomedical Sciences Inc. (EMBSI) support ExxonMobil’s research efforts for health and environmental science, including biodegradability and aquatic toxicity. The EMBSI environmental laboratory is tailored to evaluate petrochemicals in biological systems and perform various environmental toxicity tests. EMBSI is at the forefront of developing test methods for low solubility substances, such as the dietary bioaccumulation test adopted by OECD.
Our Product Stewardship & Regulatory Affairs (PS&RA) team can also provide product safety expertise and advice on regulations that may impact the markets relevant to our products.
Exxal alcohols are isomeric branched, primary alcohols that contain both even- and odd-numbered hydrocarbon chains, ranging from C8 to C13. In general, there are two main types of structures in industrial alcohols:
- Linear (or straight-chain) molecules: obtained from synthetic or natural sources;
- Branched chain alcohols: produced from propylene and butene.
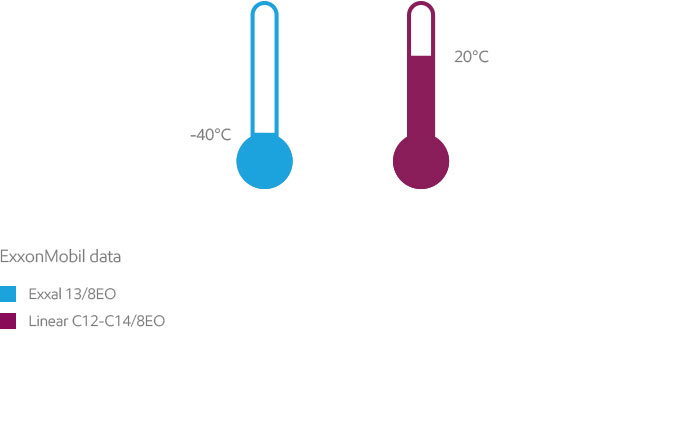
High-purity Exxal alcohols exhibit reactivity typical of higher primary alcohols. Thanks to their branched structure, Exxal alcohols are characterized by low pour points.
The pour point is the temperature below which the product does not flow. A low pour point facilitates transportation, storage and handling of products at low temperatures. While linear C12-C14 alcohol has a pour point around room temperature (20°C), branched alcohols such as Exxal 13 (an iso-tridecyl alcohol) have a pour point lower than -40 degrees. For operations in colder climates, using Exxal branched alcohols versus linear alcohols can reduce the need for heated tanks and lines. This could translate into lower energy bills and reduced handling costs, potentially leading to significant cost savings.